Chemistry
Chemistry occupies a pivotal scale in the UFD framework. It is the level at which nuclear geometry, field resonance, and harmonic constraints converge to produce stable atoms and molecules. The patterns described in quantum mechanics—orbital structure, exclusion rules, shell filling, and periodic behavior—are understood here as expressions of geometric coherence across nested fields.
At this scale, the structured nucleus establishes boundary conditions in the surrounding field, shaping the standing-wave resonances that electrons inhabit. These resonances determine atomic identity and guide how atoms interact. When atomic standing waves merge, transfer, or dissolve into shared configurations, chemical bonds form. Covalent, ionic, and metallic bonds represent distinct ways in which resonant stability is distributed across systems.
The periodic table emerges from this layered coherence. As atomic number increases, the depth and geometry of the nuclear potential well evolve, allowing new orbital resonances to become admissible. Chemical properties follow directly from the stability and accessibility of these outer resonances.
The sections that follow move from nuclear coherence and the Alpha Stability Index to atomic orbitals and bonding, and finally to the periodic organization of the elements. Together, they present chemistry as the study of how matter arranges itself into increasingly stable and expressive resonant forms within a structured plenum.
The Platonic Nucleus
Standard Model View
In conventional nuclear physics, nuclei are composed of protons and neutrons bound by the strong force. The shell model describes nucleons as occupying quantized energy levels within a mean potential well. Magic numbers arise from closed shells, and nuclear stability is associated with filled energy levels. While the model predicts many stability trends, it does not assign a specific geometric structure to most nuclei beyond probabilistic density distributions.
UFD View
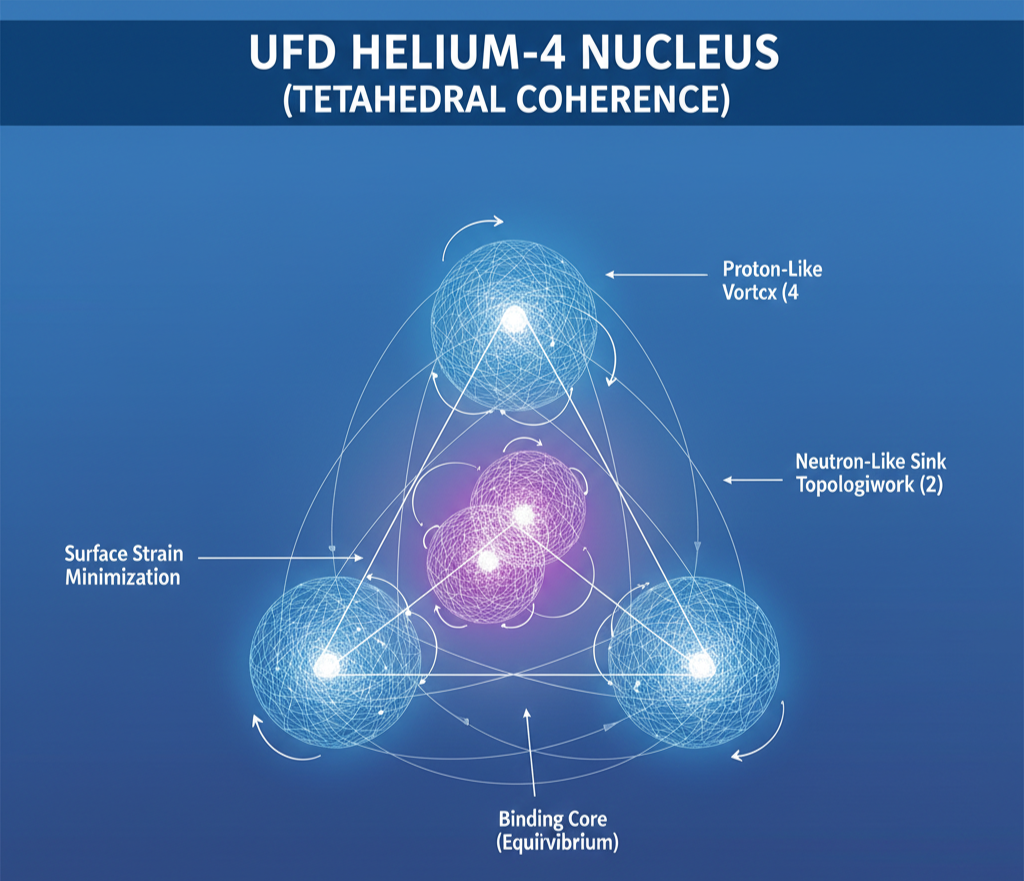
In UFD, the nucleus is a vortex lattice embedded in the Universal Energetic Field (UEF). Protons and neutrons are not point particles but coherent topological vortices whose mutual packing determines stability. Nuclear structure therefore reflects geometric efficiency: configurations that minimize residual strain in the vortex lattice correspond to deeper coherence basins.
Highly stable nuclei correspond to symmetric geometric shells—often approximating Platonic solids, such as the tetrahedral nucleus of Helium-4. These configurations represent optimal three-dimensional packing of vortex structures within a finite-coherence medium. Stability is not imposed by abstract forces but emerges from surface-strain minimization and topological locking within the UEF.
The Alpha Stability Index (ASI)
Standard Model View
Nuclear stability is typically evaluated using binding energy per nucleon. Peaks in binding energy, such as around iron-56, indicate particularly stable nuclei. The alpha cluster model further observes that many nuclei behave as if composed of helium-4 subunits, but this is treated as a phenomenological pattern rather than a geometrically grounded principle. Stability trends are modeled using semi-empirical mass formulas and shell corrections, without assigning a fundamental geometric building block to nuclear matter.
UFD View
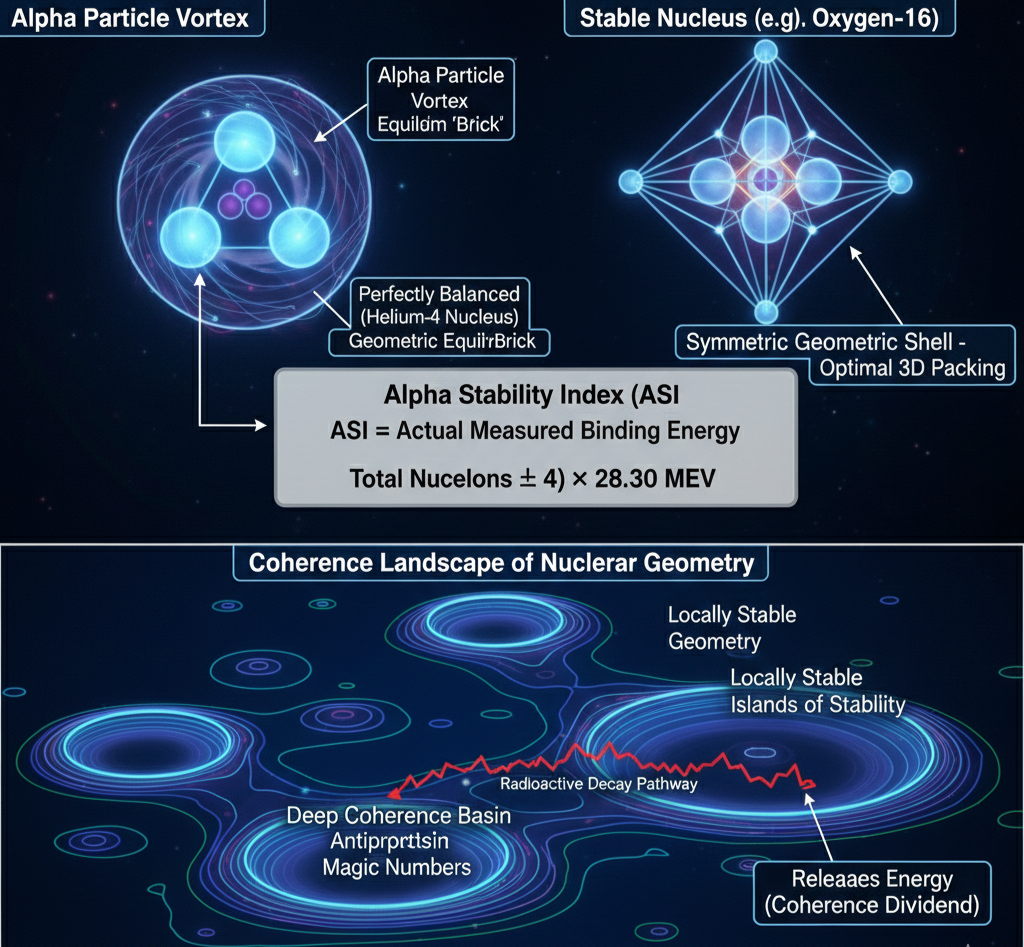
In UFD, the Alpha Stability Index (ASI) is a quantitative tool for measuring how closely a nucleus adheres to geometric coherence. The foundational unit of nuclear stability is the helium-4 nucleus—the alpha particle. In this framework, its constituents are four proton “source” vortices arranged in a tetrahedral configuration and two antiproton “sink” vortices at the geometric center. This configuration creates a perfectly balanced dynamic equilibrium, producing an exceptionally deep coherence basin. The alpha vortex is therefore the fundamental geometric “brick” of nuclear matter.
The ASI measures stability by comparing a nucleus’s actual measured binding energy to the binding energy it would possess if it were ideally composed of perfectly coherent alpha units. It is defined as:
ASI = (Actual Measured Binding Energy) / (Ideal Geometric Binding Energy)
where the ideal binding energy is calculated as:
Ideal Geometric Binding Energy = (Total Nucleons ÷ 4) × 28.30 MeV
An ASI at or above 1.0 indicates that the nucleus is at least as coherent as a simple collection of alpha vortices, and may even possess an additional coherence dividend from synergistic packing among those alpha units. Values below 1.0 indicate geometric strain. However, absolute magnitude alone does not determine stability. What matters is position within the coherence landscape. A nucleus may sit in a local basin yet remain globally unstable if a deeper configuration exists. Radioactive decay, in this framework, is the transition from a locally coherent geometry to a more globally optimal one, releasing the difference in coherence dividend as energy. The ASI thus serves not merely as a threshold measure but as a topographic map of nuclear geometry, revealing magic numbers, islands of stability, and predictable decay pathways as features of a structured coherence terrain.
Atomic Orbitals
Standard Model View
Atomic orbitals are solutions to the Schrödinger equation for electrons in a Coulomb potential. They represent probability distributions describing where an electron is likely to be found. The shapes of s, p, d, and f orbitals arise mathematically from angular momentum quantization and boundary conditions.
UFD View
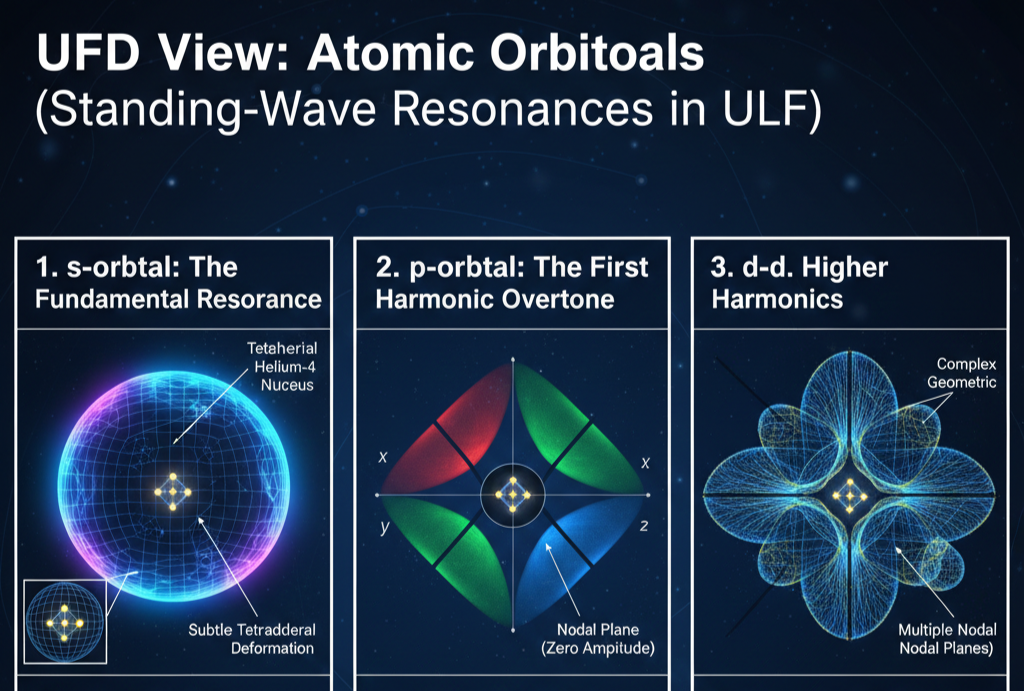
In UFD, atomic orbitals are standing-wave resonances in the Universal Light Field (ULF). The nucleus provides the central boundary condition, and only certain harmonic modes can be sustained in the surrounding medium. Orbital shapes arise from geometric admissibility in three dimensions, not from abstract probability amplitudes.
The familiar s, p, d, and f patterns reflect distinct angular harmonics of the ULF around a structured nucleus. The electron is a stable toroidal vortex phase-locked into one of these resonant modes. The orbital is not a statistical cloud but a real field configuration.
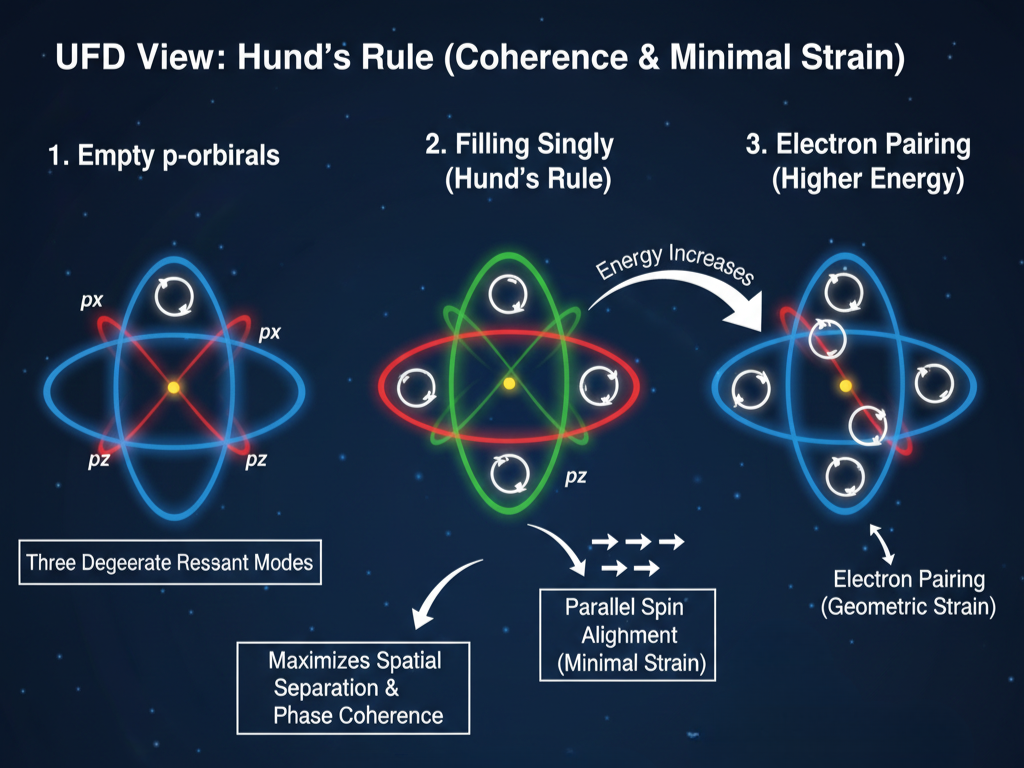
Hund’s Rule
Standard Model View
Hund’s rule states that electrons fill degenerate orbitals singly with parallel spins before pairing occurs. This minimizes electron–electron repulsion and lowers total energy. The rule is derived from quantum exchange symmetry and Coulomb interactions.
UFD View
In UFD, degenerate orbitals represent multiple equivalent resonant modes. Filling them singly maximizes spatial separation and preserves phase coherence in the ULF. Parallel alignment corresponds to a configuration that minimizes destructive interference between circulating electron vortices.
Electron pairing only occurs when lower-energy geometric options are exhausted. Hund’s rule reflects the system’s tendency to maintain maximal coherence and minimal mutual strain within the resonance structure.
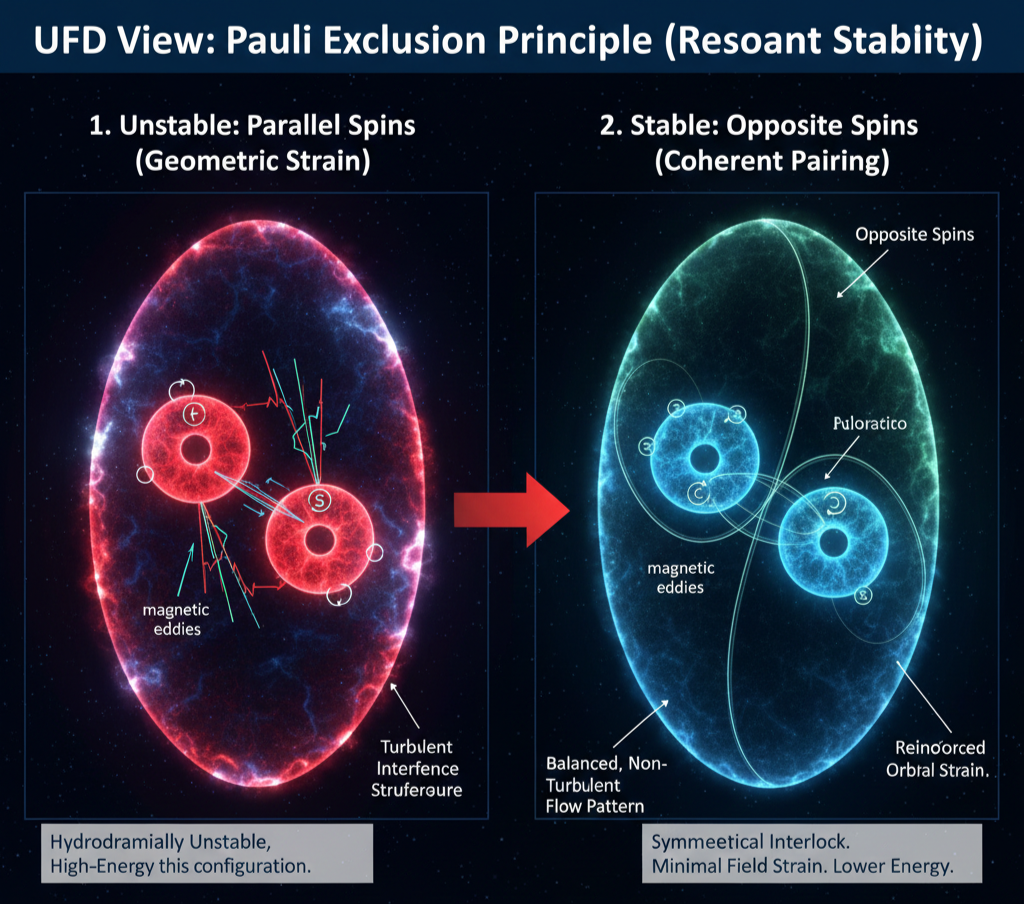
The Pauli Exclusion Principle
Standard Model View
The Pauli Exclusion Principle states that no two identical fermions can occupy the same quantum state simultaneously. In quantum mechanics, this arises from the antisymmetric nature of the fermionic wavefunction: exchanging two electrons introduces a sign change, which mathematically forbids identical state occupancy. The principle explains atomic shell structure, electron pairing, degeneracy pressure, and the stability of matter. However, its origin is treated as a fundamental algebraic property of quantum statistics rather than a physical mechanism.
UFD View
In UFD, exclusion arises from the Principle of Resonant Stability within the Universal Light Field (ULF). An atomic orbital is a standing-wave resonance, and electrons are toroidal vortices embedded within that resonance. Each electron carries a circulating magnetic eddy associated with its spin. When two electrons with parallel spins attempt to occupy the same orbital, their magnetic eddies rotate in the same direction. This produces turbulent interference in the surrounding ULF, disrupting the orbital’s standing-wave structure and generating a hydrodynamically unstable, high-energy configuration.
Stability is only achieved when two electrons occupy the same orbital with opposite spins. In this case, their counter-rotating magnetic eddies interlock and mesh symmetrically, creating a balanced, non-turbulent flow pattern that reinforces the orbital resonance. This paired configuration minimizes field strain and lowers the system’s energy. The orbital can therefore support at most two electrons, provided their spins are opposite. In this framework, the Pauli Exclusion Principle is not an abstract statistical rule but a physical constraint arising from the hydrodynamic coherence requirements of real field structures.
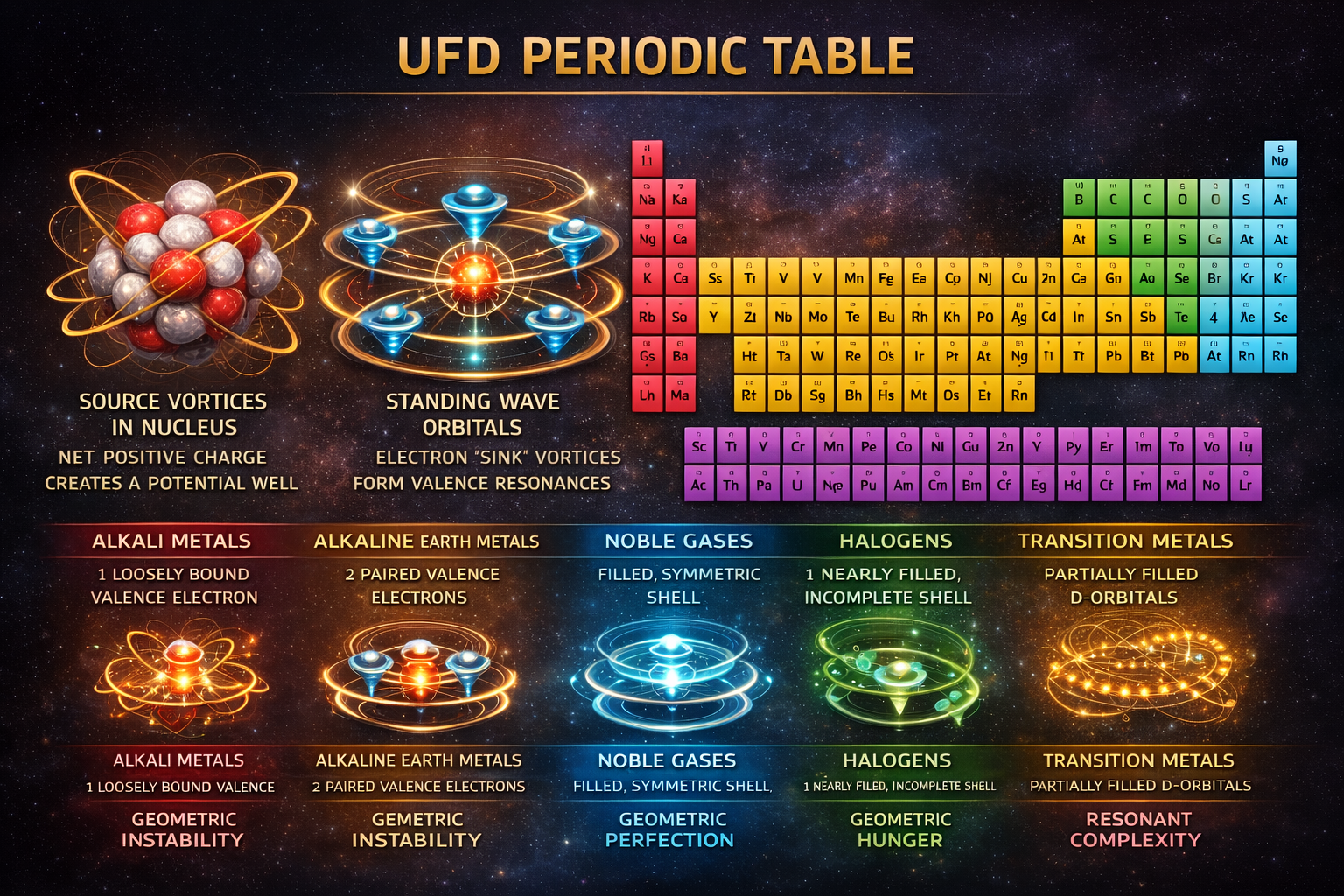
The Periodic Table
Standard Model View
The periodic table organizes elements by increasing atomic number (Z), which corresponds to the number of protons in the nucleus. Chemical periodicity arises from electron shell structure: as atomic number increases, electrons fill quantized orbitals in a repeating pattern. Elements in the same column share similar valence electron configurations, which gives rise to recurring chemical properties across periods. The table is therefore understood as a consequence of quantum mechanical shell filling and electrostatic attraction between electrons and the nucleus.
UFD View
In UFD, the atomic number represents the number of un-neutralized UEF “source” vortices in the nucleus. Each proton contributes an outward source flow in the Universal Energetic Field, generating a net positive charge. This net source count creates a structured potential well in the surrounding Universal Light Field (ULF). The depth and geometry of this well determine the number and shape of the stable standing-wave resonances that electron “sink” vortices must form to reach equilibrium.
The periodic table therefore reflects the progressive construction of stable resonant shells in the ULF. As Z increases, the potential well strengthens, enabling new orbital geometries to become admissible. Chemical behavior is not determined merely by electron count, but by the stability and geometric accessibility of the outermost standing wave—the valence resonance. Elements in the same group share similar outer resonance structures, which is why they exhibit analogous chemical properties. In this framework, the periodic table is a map of resonant equilibrium states generated by increasing net source strength within a structured plenum.
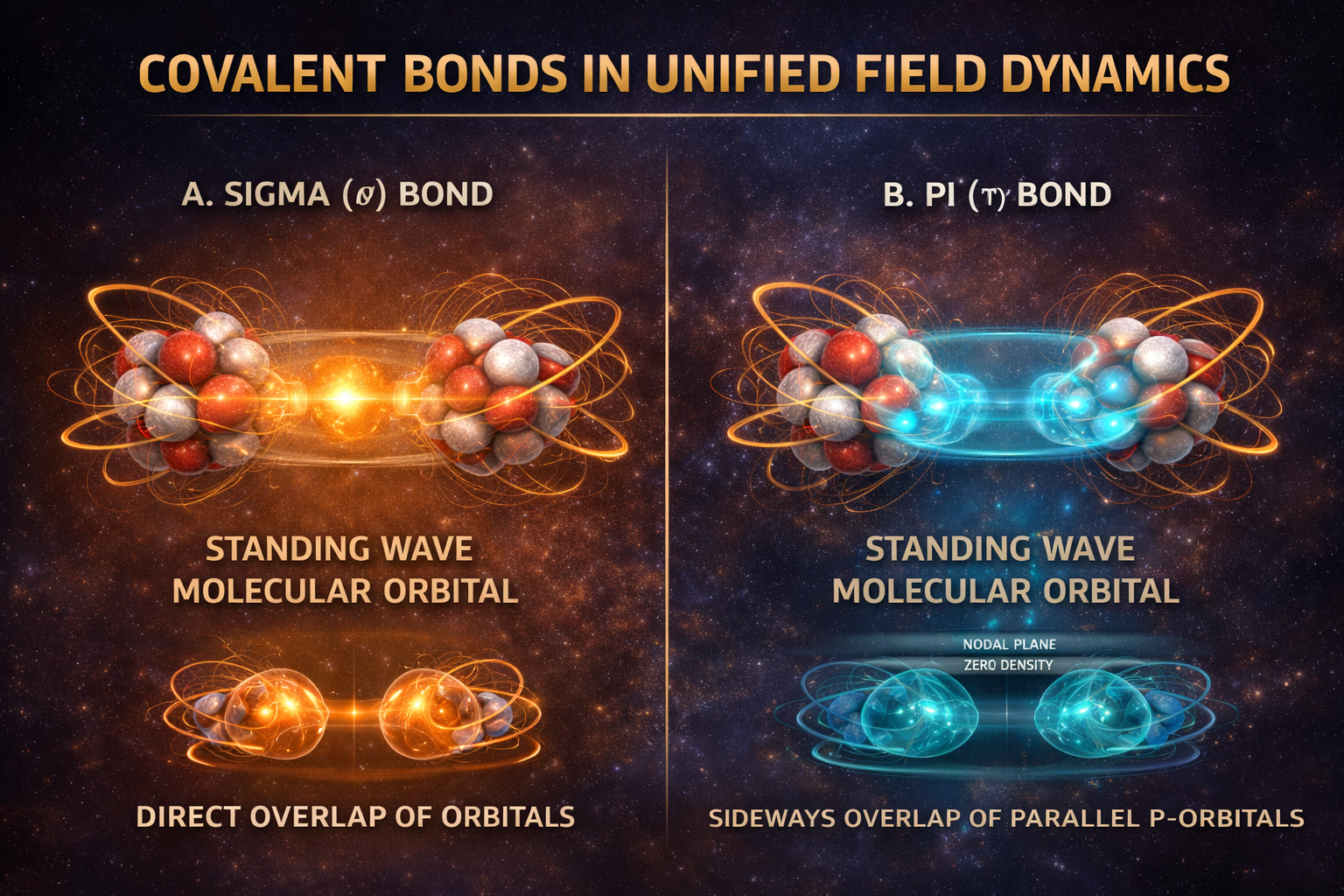
Covalent Bonds
Standard Model View
In conventional chemistry, a covalent bond forms when two atoms share one or more electrons. According to valence bond theory and molecular orbital theory, atomic orbitals overlap to create a lower-energy molecular orbital that extends over both nuclei. The electrons occupying this shared orbital lower the total energy of the system, producing an attractive force that binds the atoms together. The nucleus is typically treated as a stationary positive point charge, and bond strength is determined primarily by electron distribution and orbital overlap.
UFD View
In UFD, a covalent bond is a two-level resonance governed by the Principle of Geometric Congruence. At the first level—the UEF foundation—the interacting nuclei are not passive point charges but structured vortex assemblies. Bond strength depends in part on whether the underlying nuclear geometries can align or “mesh” coherently. When two nuclear vortex structures achieve geometric compatibility, they form a more stable energetic foundation. This nuclear congruence stabilizes the entire bonded configuration at the deepest structural level.
At the second level—the ULF superstructure—the bond forms when two atomic standing waves merge into a single, shared resonance encompassing both nuclei. This merged wave is the molecular orbital. When the system transitions into this more coherent configuration, it releases a Resonance Dividend—the molecular analogue of the nuclear Coherence Dividend. A sigma (σ) bond forms through direct, end-to-end orbital overlap, producing a cylindrically symmetric standing wave concentrated along the internuclear axis. A pi (π) bond forms through sideways overlap of parallel p-orbitals, generating two lobed regions of shared resonance above and below the axis with a nodal plane between them. Molecular structure and bond strength are therefore determined by how efficiently these shared standing waves achieve geometric and hydrodynamic coherence across both fields.
Ionic Bonds
Standard Model View
An ionic bond forms when one atom transfers one or more electrons to another atom. This typically occurs between a metal, which tends to lose electrons, and a nonmetal, which tends to gain them. The transfer creates oppositely charged ions: a positively charged cation and a negatively charged anion. The electrostatic attraction between these oppositely charged ions forms the bond. Bond strength is largely determined by Coulombic attraction and lattice energy in the resulting ionic solid.
UFD View
In UFD, the ionic bond is a transfer of resonant stability. The process begins when one atomic system is geometrically unstable, possessing a loosely bound electron vortex in a high-energy outer resonance, while a neighboring atom contains a lower-energy, geometrically admissible resonance that remains unoccupied. The system as a whole can lower its total energy when the electron vortex abandons the unstable standing wave of the first atom and relocalizes into the more stable resonance of the second. This physical relocation of a vortex within the ULF is the electron transfer.
The consequence of this transfer is the creation of two new field asymmetries. The atom that loses the electron now has an excess of unneutralized UEF “source” potential and becomes a cation. The atom that gains the electron acquires excess “sink” potential and becomes an anion. These complementary imbalances generate a fluid-dynamic flow in the surrounding fields, drawing the ions together through source–sink coupling. The strength of the ionic bond is determined by the net Resonance Dividend released when the electron settles into its new, more coherent state. Ionic bonding, therefore, is not merely electrostatic attraction but the system-wide migration toward maximal geometric coherence across both fields.
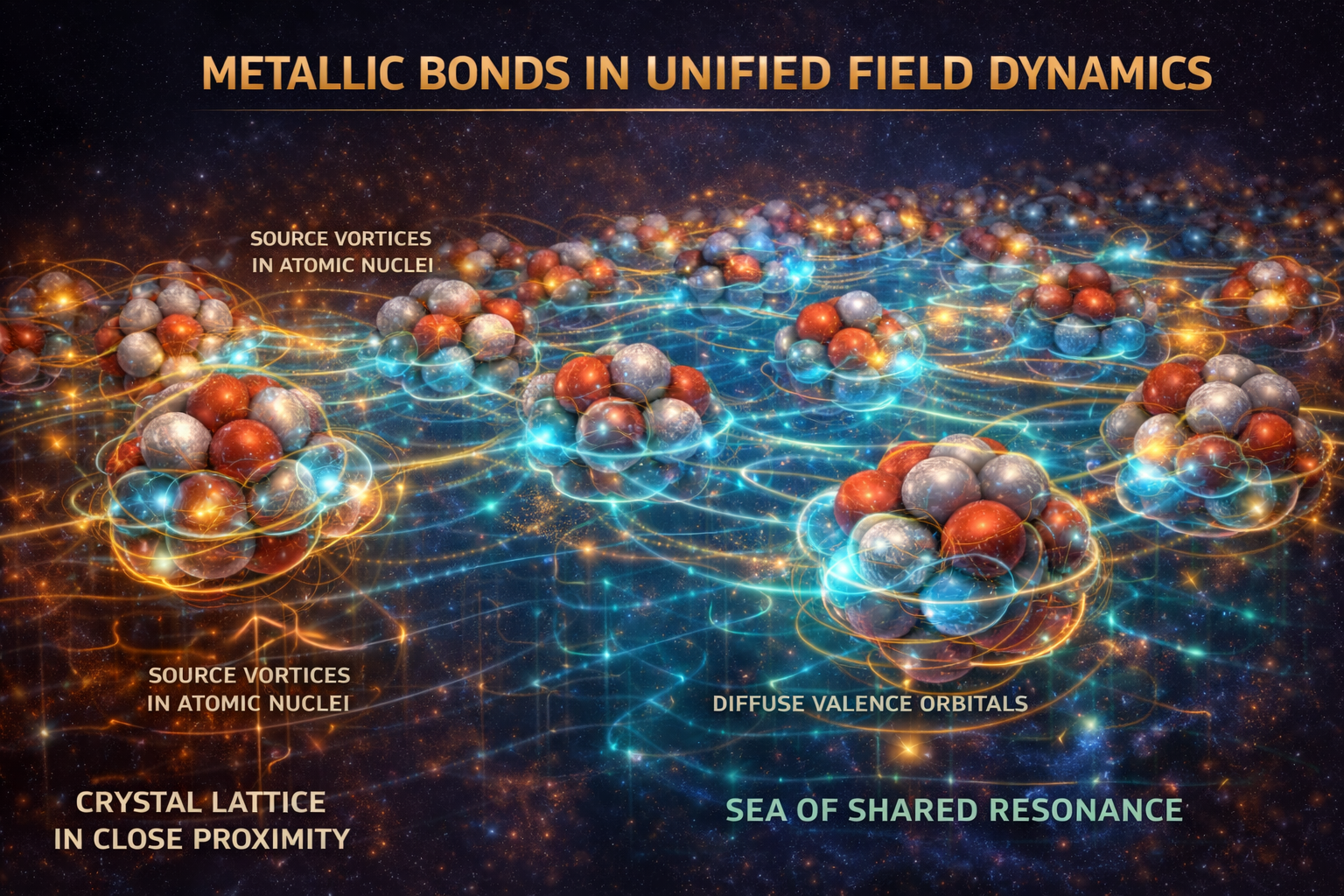
Metallic Bonds
Standard Model View
In conventional chemistry and solid-state physics, a metallic bond is described as a lattice of positively charged metal ions immersed in a “sea of delocalized electrons.” These electrons are not bound to any one atom but are free to move throughout the crystal. This delocalization explains the defining properties of metals, including high electrical and thermal conductivity, metallic luster, and malleability. The bonding is therefore understood as collective electrostatic attraction between ion cores and mobile conduction electrons.
UFD View
In UFD, the metallic bond is an emergent state of shared ULF resonance. It arises from two key structural conditions common to metals: efficient crystal packing and diffuse valence orbitals. Metal atoms arrange themselves in tightly packed lattices, minimizing core separation. At the same time, their outer electron vortices occupy large, weakly bound standing waves—especially in the d- and f-orbitals of transition and inner-transition metals. These diffuse resonances extend far from the nucleus.
When such atoms are brought into close proximity, their expansive ULF standing waves overlap extensively. No single nucleus can maintain exclusive control over its valence electron vortices. The individual atomic orbitals therefore lose their independence and merge into a single, crystal-wide standing wave—a continuous sea of shared resonance. This collective ULF structure allows electron vortices to flow freely across the lattice, explaining electrical and thermal conductivity. The surface of this shared resonance readily absorbs and re-emits photons, producing metallic luster. Because cohesion is maintained by a fluid-like, distributed resonance rather than rigid directional bonds, atomic cores can slide past one another without breaking the underlying coherence, giving rise to malleability and ductility. The metallic bond is thus not merely delocalized charge, but a macroscopic standing-wave state spanning the entire crystal.
*Images were generated with the assistance of Gemini and ChatGPT