Neuroscience
Neuroscience has traditionally described the brain as a biochemical and electrical network composed of neurons that communicate through synaptic signaling and action potentials. Over the past century, this framework has mapped neural circuits, identified neurotransmitter systems, and revealed the importance of oscillatory rhythms and large-scale networks in shaping perception, memory, and behavior. Modern imaging and electrophysiology now show that cognition depends not only on individual neurons, but on dynamic coordination across distributed brain systems.
Yet fundamental questions remain. While neural correlates of consciousness can be identified, the physical basis of subjective experience, intentionality, and unified awareness remains unresolved. The brain appears to operate as an integrated whole, but classical mechanistic models struggle to explain how coherence across scales gives rise to unified mental states.
Resonant Neuroscience (RN), the application of Unified Field Dynamics (UFD) to brain science, reframes the brain as a hierarchically coherent field system. In this view, neurons function as oscillatory waveguides, neurotransmitters as geometric modulators, and large-scale networks as dynamically coupled resonance structures. Cognitive processes emerge from nested phase alignment across these interacting geometries. Rather than reducing mind to computation alone, RN proposes that thought, perception, and awareness arise from coordinated resonance within and across neural fields.
This section explores the geometry of neurons, the dynamics of brain rhythms, the organization of large-scale networks, and the deeper field interactions that may underlie conscious experience. By integrating neurophysiology with field theory and resonance dynamics, it offers a unified perspective on how energy becomes mind.
The Neuron
Standard Model View
The neuron is the fundamental signaling unit of the nervous system. It consists of dendrites, which receive synaptic input; a cell body (soma), which integrates electrical signals; and an axon, which transmits action potentials to other cells. Neural communication occurs through voltage-gated ion channels that generate electrical impulses along the axon, followed by neurotransmitter release at synapses. Myelination increases conduction speed by enabling saltatory transmission between nodes of Ranvier. In this framework, neurons function as electrochemical signal processors within a network of connected cells.
UFD View
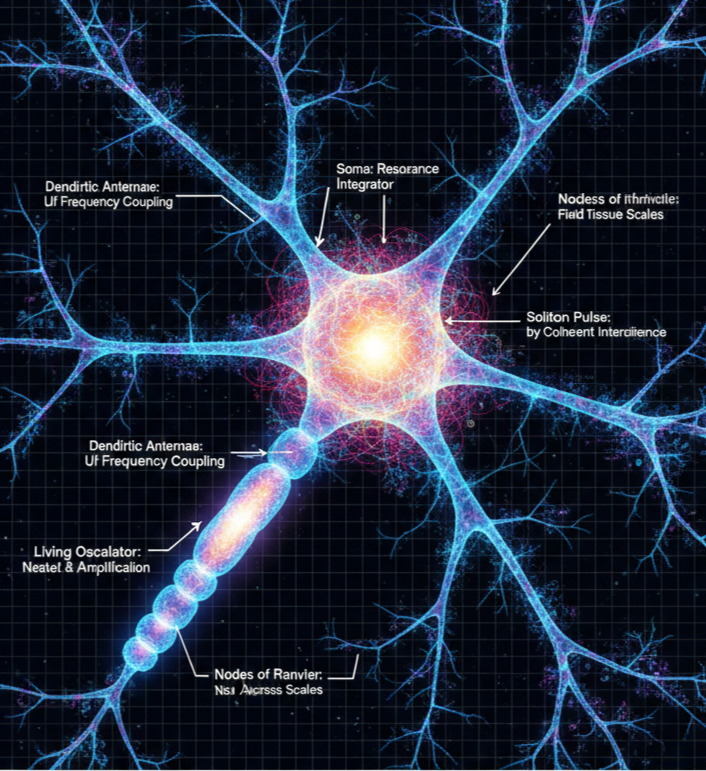
In UFD, the neuron is a resonant structure embedded within a coherent field environment. Its morphology is not merely anatomical but geometrically tuned for vibrational integration and transmission. Dendritic branching patterns act as frequency-sensitive antennae that couple to ultra-light-frequency (Ulf) standing wave activity in surrounding neural tissue. The soma functions as a resonance integrator, summing incoming vibrational inputs and determining phase-alignment thresholds for coherent propagation.
The axon operates as a cylindrical waveguide that supports soliton-like pulses sustained by constructive interference between membrane potentials and cytoskeletal field geometry. Myelination enhances transmission fidelity by stabilizing phase continuity, while nodes of Ranvier serve as periodic phase-reset and amplification points. In this view, neurons are not merely biological wires but living oscillators participating in recursive field entrainment and nested coherence across neural scales.
Action Potentials
Standard Model View
An action potential is a rapid, transient change in membrane voltage that propagates along the axon. When depolarization reaches a threshold, voltage-gated ion channels open, allowing sodium ions to enter the neuron, followed by potassium efflux that restores the resting potential. This sequence produces a self-propagating electrical impulse governed by ion gradients and membrane permeability. The signal is “all-or-none,” meaning that once threshold is reached, the full spike is generated and travels without decrement along the axon.
UFD View
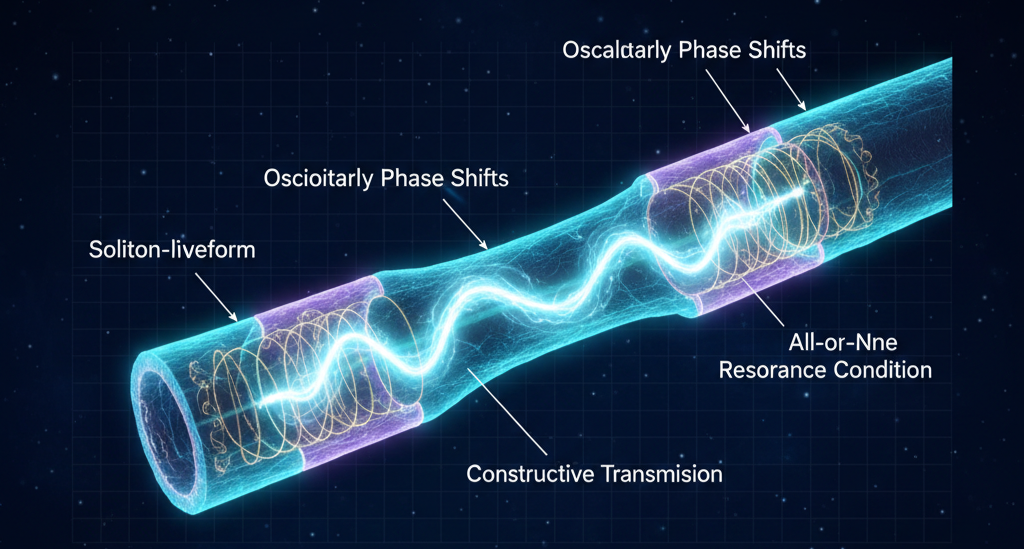
In UFD, the action potential is a phase-guided resonance pulse rather than merely a voltage spike. It functions as a soliton-like waveform that maintains structural coherence as it travels along the axonal waveguide. Depolarization and repolarization correspond to oscillatory phase shifts in the membrane’s field geometry, transforming ionic gradients into coordinated energy exchanges across the membrane and cytoskeletal lattice.
Propagation depends on constructive interference between the membrane’s standing-wave structure and deeper cytoskeletal resonances. The “all-or-none” character reflects a binary resonance condition: the pulse either phase-locks with the axonal geometry and sustains coherent transmission, or it fails to align and collapses. In this view, neural firing preserves information through waveform continuity, not merely electrical amplitude, allowing action potentials to function as coherent signaling events within a nested field architecture.
Neurotransmitters
Standard Model View
Neurotransmitters are chemical messengers released from presynaptic neurons that bind to receptors on postsynaptic cells. Their effects depend on receptor type and ion-channel modulation, producing excitatory or inhibitory responses. Glutamate is the primary excitatory neurotransmitter in the brain, promoting depolarization and increasing the likelihood of action potentials. GABA and glycine serve inhibitory roles, reducing neuronal firing by increasing membrane permeability to chloride or potassium ions. The balance between excitation and inhibition regulates neural rhythms, prevents runaway activity, and supports cognitive processes such as attention, memory, and perception.
UFD View
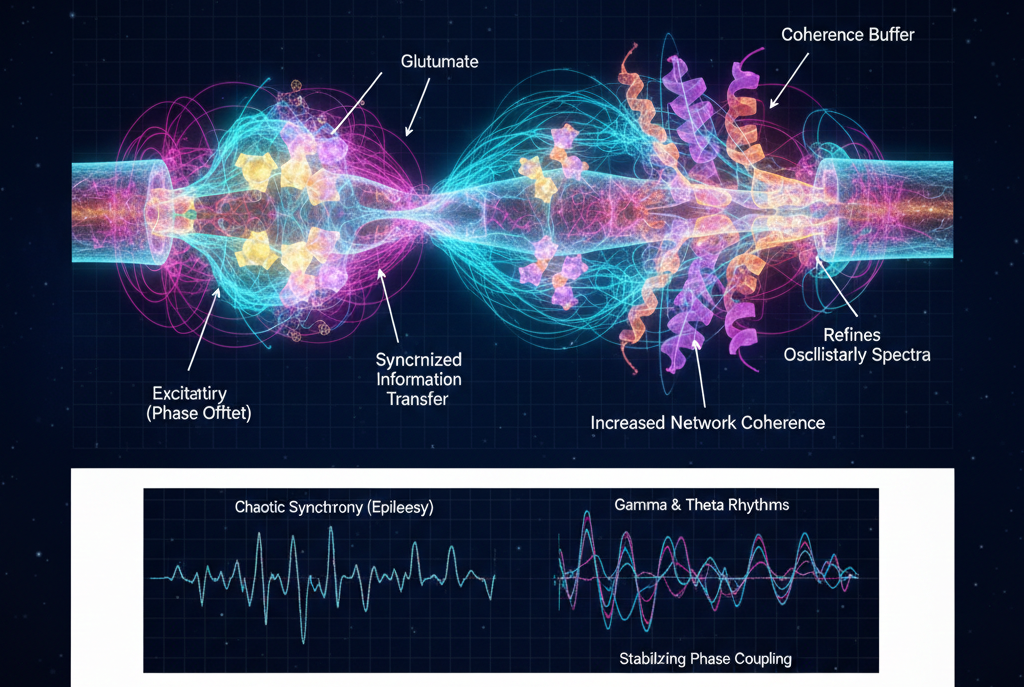
In UFD, neurotransmitters function as modulators of resonance rather than merely chemical carriers. Their molecular geometry determines how they phase-align with synaptic receptors and the surrounding neural field. Binding events act as moments of geometric entrainment, shifting the local oscillatory state of the neuron and influencing broader network coherence.
Excitatory neurotransmitters promote phase amplification. Glutamate enhances local resonance alignment, increasing coherence and facilitating synchronized information transfer. Inhibitory neurotransmitters introduce stabilizing phase offsets. GABA and glycine act as coherence buffers, refining oscillatory spectra and preventing destructive synchronization. Rather than simply silencing neurons, inhibitory modulation sculpts neural rhythms into stable, functional patterns.
The interplay between excitation and inhibition represents a resonant trade-off. Healthy brain function depends on maintaining structured phase balance across scales. When inhibitory buffering fails, as in epilepsy, coherence collapses into chaotic synchrony. When excitation and inhibition are precisely timed, gamma and theta rhythms emerge from controlled phase coupling. In this view, neurotransmitters are geometric keys that unlock and reshape resonant states within nested neural field architectures.
Microtubules
Standard Model View
Microtubules are cylindrical polymers composed of tubulin dimers that form part of the cytoskeleton. They provide structural support, maintain cell shape, facilitate intracellular transport, and play essential roles in mitosis and axonal trafficking. In neurons, microtubules help organize axonal structure and enable motor proteins to transport vesicles and organelles between the soma and synaptic terminals. Their function is typically understood in mechanical and logistical terms rather than in informational or integrative roles.
UFD View
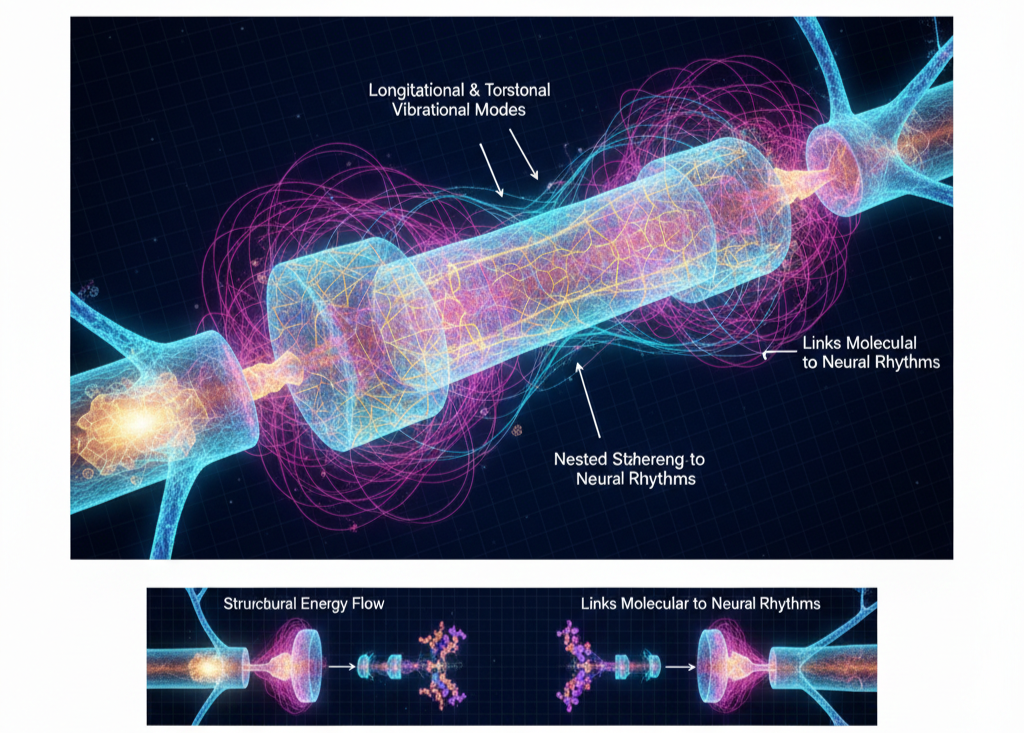
In UFD, microtubules function as intracellular resonance conduits rather than merely structural scaffolding. Their quasi-crystalline cylindrical lattice supports longitudinal and torsional vibrational modes, enabling nested standing-wave patterns within the neuron. These oscillations, sustained by tubulin dynamics and intracellular energy flow, allow spatially separated regions of the neuron to synchronize through phase alignment.
Because of their ordered geometry and dielectric properties, microtubules are well suited to act as waveguides for ultra-light-frequency (Ulf) harmonics. They provide the geometric backbone that links molecular signaling to larger-scale neural rhythms, enabling distributed coherence across dendrites, soma, and axon. Structural reconfiguration of the tubulin lattice may modulate a neuron’s resonant profile, suggesting a potential deeper substrate for memory beyond synaptic strength alone. In this view, microtubules bridge molecular processes with mesoscale neural dynamics, supporting nested coherence across scales.
Neutral Coherence
Standard Model View
Modern neuroscience recognizes that brain function depends on coordinated oscillatory activity across multiple spatial and temporal scales. Neural rhythms—commonly categorized into delta, theta, alpha, beta, and gamma frequency bands—are associated with distinct cognitive and physiological states. Cross-frequency coupling allows slower rhythms to modulate faster ones, enabling large-scale integration and local processing to occur simultaneously. Functional brain networks such as the Default Mode Network (DMN), Central Executive Network (CEN), and Salience Network (SN) are understood as dynamically interacting systems that coordinate attention, memory, introspection, and task engagement.
In this framework, coherence refers to synchronized neural firing across regions, supporting efficient information transfer. Cognitive clarity arises from well-timed synchronization, while disorders such as epilepsy, schizophrenia, and neurodegeneration are associated with abnormal oscillatory patterns and disrupted network coordination.
UFD View
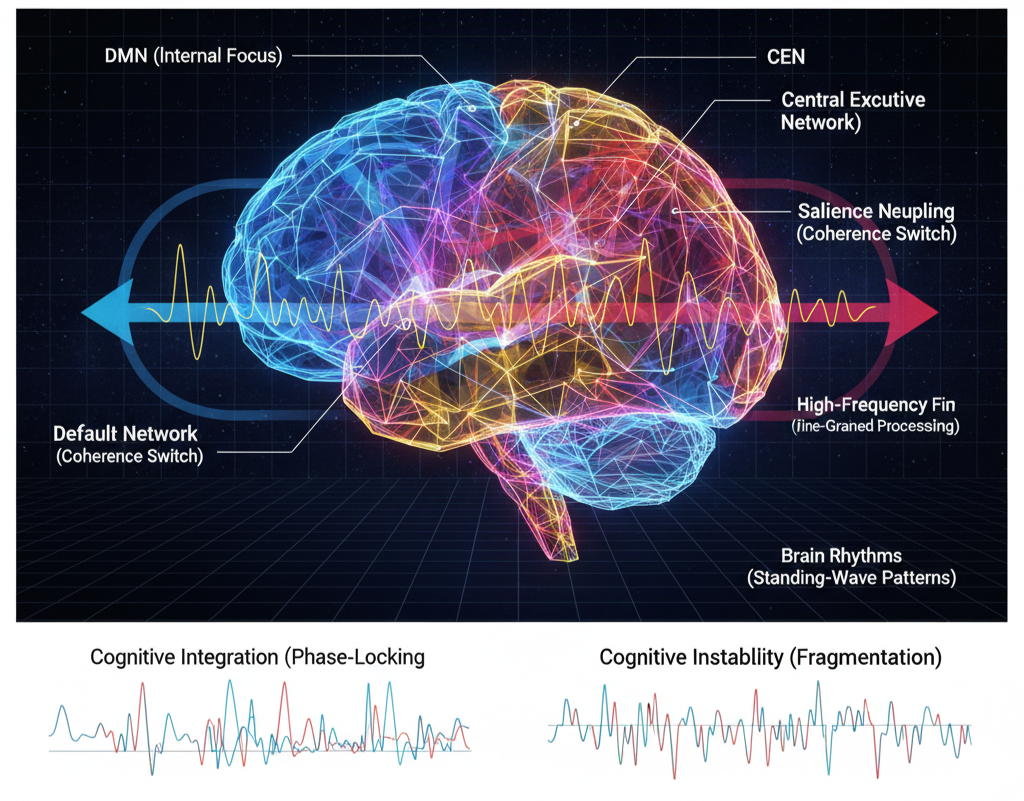
In UFD, neural coherence is not merely correlated activity but structured field alignment across nested oscillatory hierarchies. Brain rhythms are standing-wave patterns stabilized by the geometry of neural tissue and its electromagnetic field architecture. Lower-frequency oscillations serve as large-scale phase envelopes, while higher-frequency rhythms coordinate fine-grained processing within those envelopes. Cross-frequency coupling reflects nested resonance relationships rather than independent channels of activity.
Large-scale networks such as the DMN, CEN, and SN represent stable attractor modes within the brain’s resonant field space. Transitions between networks correspond to phase redistributions across this geometry. The Salience Network functions as a coherence switch, modulating phase alignment between internally oriented and externally directed states.
Cognitive integration emerges when oscillatory layers phase-lock across scales, producing unified experiential states. Fragmentation of coherence leads to cognitive instability, attentional breakdown, or pathological synchrony. In this view, the brain operates as a dynamic resonance spectrum, where mental states correspond to structured configurations of nested field alignment.
The Coherence Project
Standard Model View
Neurological and psychiatric disorders are typically understood as disruptions in neural circuitry, neurotransmitter balance, genetic regulation, or structural integrity. Epilepsy involves excessive synchronous firing; schizophrenia is associated with disrupted connectivity and abnormal oscillations; Alzheimer’s disease reflects progressive neurodegeneration; mood and attention disorders involve dysregulated neurotransmitter systems and network imbalances. Treatments generally target chemical pathways, electrical excitability, or structural decline through pharmacology, stimulation, or behavioral therapy.
UFD View
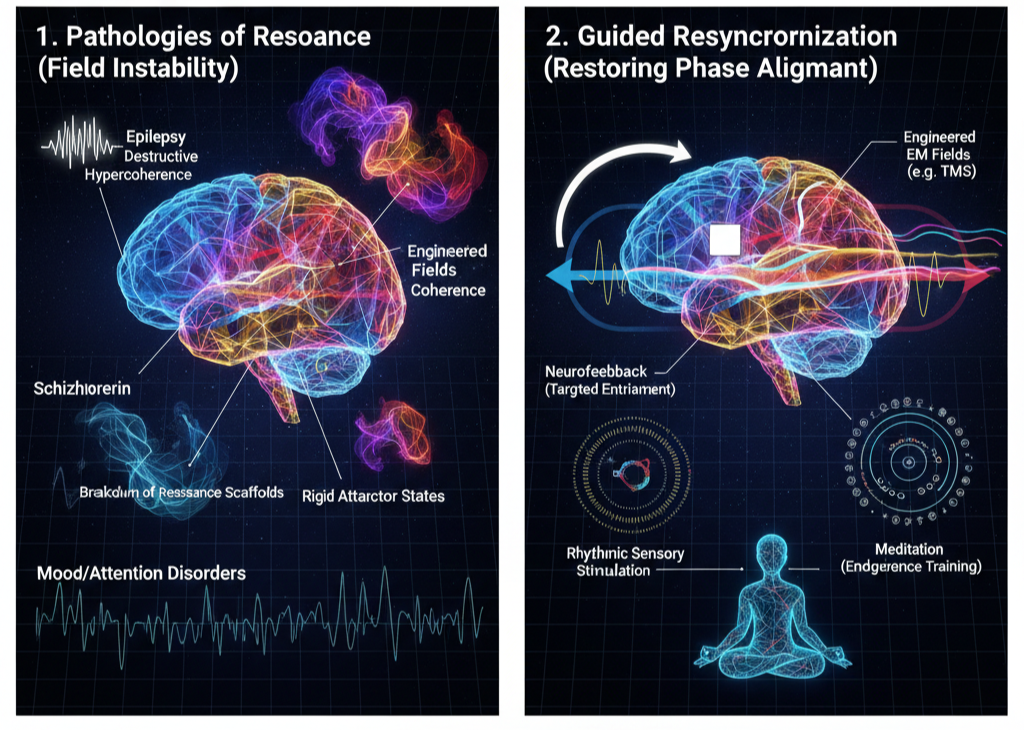
In UFD, these conditions can be reinterpreted as pathologies of resonance—failures in the brain’s ability to maintain coherent phase relationships across nested oscillatory hierarchies. Epilepsy reflects destructive hypercoherence, where low-complexity synchronization overwhelms dynamic flexibility. Schizophrenia represents fragmented coherence, in which local and global oscillatory layers decouple. Neurodegenerative disorders correspond to gradual breakdowns of structural resonance scaffolds that support memory and integration. Mood and attention disorders emerge from rigid or unstable attractor states within the brain’s resonance spectrum.
The Coherence Project reframes therapy as the restoration of phase alignment rather than merely chemical correction. Existing tools such as transcranial magnetic stimulation (TMS), neurofeedback, and rhythmic sensory stimulation already operate on this principle by externally nudging oscillatory patterns toward healthier configurations. Behavioral practices such as meditation can be understood as endogenous coherence training—intentional modulation of neural attractor states through sustained attentional entrainment.
Within this paradigm, healing involves restoring nested synchrony across scales: stabilizing local oscillations, re-establishing network integration, and re-embedding the individual within supportive environmental and relational coherence. Mental health becomes the capacity to maintain adaptive resonance under energetic flux. Disorder reflects instability in that dynamic alignment. The Coherence Project thus proposes a unifying framework in which diagnosis becomes coherence mapping and intervention becomes guided resynchronization.
*Images were created with the assistance of Gemini